Entropy change equation9/28/2023 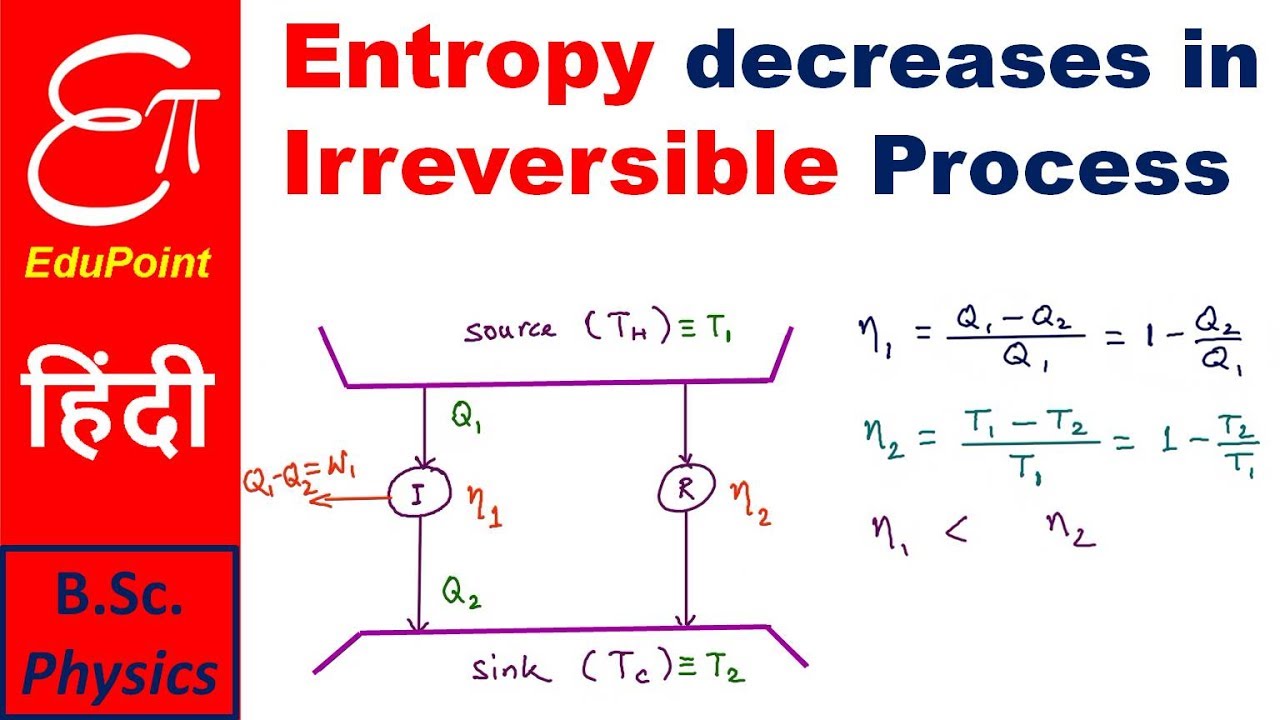
Note that the two terms on the right hand side of the equation are exact differentials. So, all one needs to do is to integrate this equation from the initial thermodynamic equilibrium state to the final thermodynamic equilibrium state determined obtained with the irreversible process. This is the same thing as a reversible path. It indicates increased randomness at the interface of solid/liquid, with some structural changes in the adsorbate and adsorbent. Positive S shows the adsorbent's affinity toward the adsorbate. Regarding the original question of why the equation for dS in terms of dT and dV for an ideal gas gives the correct answer, it is because the equation applies to the changes in dS, dT, and dV over a continuous sequence of thermodynamic equilibrium states. The entropy change (S ) during an adsorption is attained using Van't Hoff equation. I don't know why it isn't taught properly to students. You then calculate the integral of dq/T for the reversible process path. Therefore, even if the irreversible process path is adiabatic, the reversible path does not have to be. The reversible process path does not necessarily have to bear any resemblance to the actual irreversible process path. You then need to identify a reversible process path between the exact same pair of equilibrium states. In calculating the change in entropy of a closed system that has undergone an irreversible process, you need to first focus exclusively on the initial and final equilibrium states. If the divider between the filled and unfilled halves is suddenly removed, that would be a case of adiabatic, isoergic expansion. Think of gas confined to one half of a cylinder, and the other half of the cylinder is empty (i.e. $dS = 0 = \fracdV > 0$, i.e., the entropy of the gas increases, which makes sense because the process was irreversible. But keep in mind that your equation for $dS$ is only true for ideal gases. Where, Q is the heat transfer to or from the thermodynamic system. We can calculate the Entropy Change of a chemical reaction or a system by using the change in entropy formula: S (Q/T)rev. In this case you can apply both of your equations. The Entropy Change of a thermodynamic system is represented as S. Without heat flow, the only way the entropy of the universe can stay constant is if both the entropy of the universe and of the gas stay constant. If the expansion is done reversibly, then we know entropy of the universe can't be changing (because reversible), and that heat flow from the gas to the surroundings is zero (because adiabatic). The question doesn't have sufficient information for a solution, because we don't know if the adiabatic expansion is reversible or irreversible. "One mole of ideal gas initially at a pressure of 1 atmosphere and T = 298 K, is expanded into a volume 50% larger adiabatically."
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
